Cefoperazone Tazobactam Injection
Price 316.0 INR/ Unit
Cefoperazone Tazobactam Injection Specification
- Dosage Form
- As directed by physician
- Salt Composition
- Cefoperazone 1000 mg. + Tazobactam 125 mg.
- Pacakaging (Quantity Per Box)
- 1 Vial in box
- Origin of Medicine
- India
- Indication
- As directed by physician
- Drug Type
- Injection
- Ingredients
- Cefoperazone 1000 mg. + Tazobactam 125 mg.
- Physical Form
- Powder
- Function
- Antibiotic Medicine
- Recommended For
- as prescribed by physician
- Dosage
- as prescribed by physician
- Dosage Guidelines
- as prescribed by physician
- Suitable For
- Adults
- Storage Instructions
- Cool, Dry and Dark place
Cefoperazone Tazobactam Injection Trade Information
- Minimum Order Quantity
- 100 Units
- FOB Port
- Cargo, Logistic, Courer
- Payment Terms
- Cash on Delivery (COD), Cash in Advance (CID), Cheque
- Supply Ability
- 100000 Units Per Month
- Delivery Time
- 1 Days
- Sample Available
- No
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- 1 Vial in Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
- Certifications
- WHO GMP Certified
About Cefoperazone Tazobactam Injection
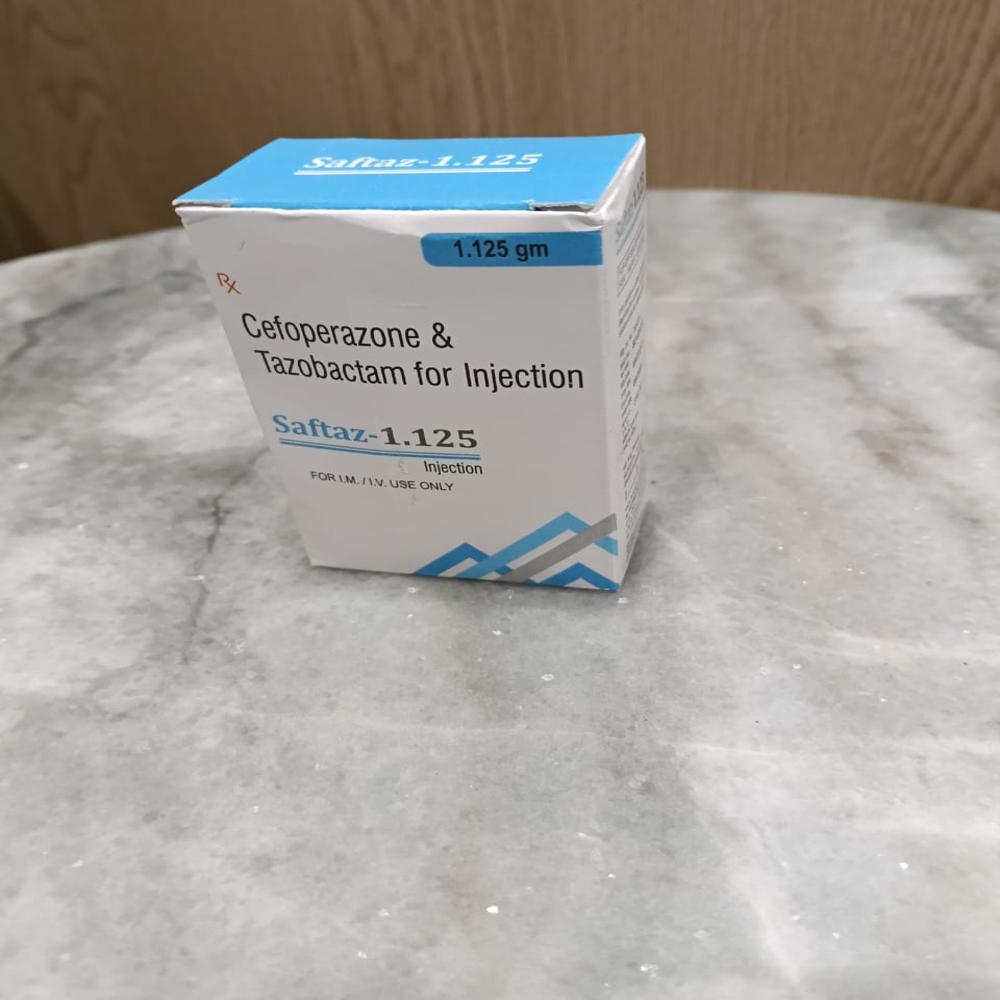
Product Name: Saftaz 1.125
Composition: Cefoperazone Sodium 1000 mg + Tazobactam Sodium 125 mg
Dosage Form: Injection (for IV/IM use)
Therapeutic Category: Third-Generation Cephalosporin with -lactamase Inhibitor
Description:
Saftaz 1.125 is a powerful combination of Cefoperazone, a third-generation cephalosporin antibiotic, and Tazobactam, a -lactamase inhibitor. Cefoperazone inhibits bacterial cell wall synthesis, while Tazobactam extends its spectrum by inhibiting -lactamase enzymes that can degrade cefoperazone. Together, they provide enhanced activity against a wide range of Gram-positive and Gram-negative bacteria, including -lactamase-producing strains.
Indications:
-
Lower respiratory tract infections
-
Urinary tract infections
-
Intra-abdominal infections
-
Septicemia
-
Skin and soft tissue infections
-
Gynecological and obstetric infections
-
Post-surgical infections
Key Benefits:
-
Broad-spectrum antibacterial coverage
-
Effective against resistant -lactamase-producing organisms
-
Suitable for serious and hospital-acquired infections
-
Reliable safety and tolerability profile
Dosage & Administration:
As prescribed by the physician. Typically administered via IV or IM route.
Powerful Dual-Action Antibiotic
This injection brings together Cefoperazone, a broad-spectrum cephalosporin antibiotic, with Tazobactam, a beta-lactamase inhibitor, amplifying its effectiveness against resistant bacteria. The formulation is particularly useful when standard antibiotics may not suffice, providing robust protection for adult patients battling severe infections.
Rigorous Quality from Indian Manufacturers
Manufactured, supplied, and distributed in India, each vial of this injection is produced under stringent quality standards. With careful sourcing and precise formulation, customers can trust the safety, purity, and reliable delivery of this essential medication for hospital and clinical use.
FAQ's of Cefoperazone Tazobactam Injection:
Q: How should Cefoperazone Tazobactam Injection be administered?
A: Cefoperazone Tazobactam Injection is administered by dissolving the dry powder in a suitable solvent and injecting it either intravenously or intramuscularly. The dosage and administration schedule should always be determined by a qualified physician.Q: What medical conditions can this injectable treat?
A: This antibiotic injection is indicated for treating various severe bacterial infections, especially those caused by organisms resistant to other antibiotics. The specific indication should be decided by a healthcare provider based on individual patient assessment.Q: When should this medication not be used?
A: Cefoperazone Tazobactam should not be used in patients with known allergies to cephalosporins, penicillins, or any component of the formulation. Always inform your physician of any allergies or prior adverse reactions.Q: Where should this product be stored to maintain its effectiveness?
A: This injection should be stored in a cool, dry, and dark place, away from direct sunlight and heat, as improper storage could compromise its potency and safety.Q: What are the key steps in the injection process?
A: A healthcare professional will dissolve the powder in an appropriate sterile solution right before administration. The solution is then injected into a vein or muscle, following standard medical protocols to ensure safety and effectiveness.Q: What are the benefits of combining Cefoperazone and Tazobactam?
A: The combination enhances antibacterial coverage, as Tazobactam inhibits enzymes that could make bacteria resistant to Cefoperazone, allowing the treatment to effectively combat a wider range of infections.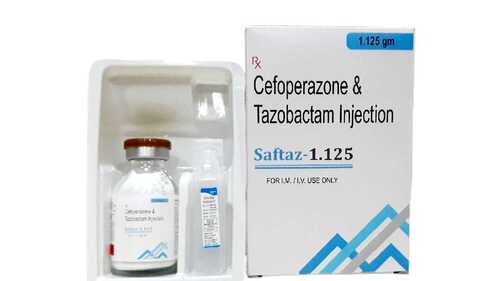





Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Antibiotic Injections Category
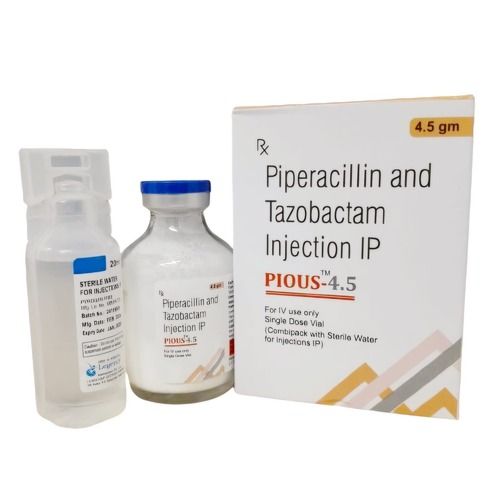
Piperacillin Tazobactam
Price 415.0 INR / Unit
Minimum Order Quantity : 100 Units
Physical Form : Powder
Ingredients : Piperacillin 4 gm+ Tazobactam 0.5 gm
Dosage Guidelines : as directed by the physician
Dosage : as directed by physician
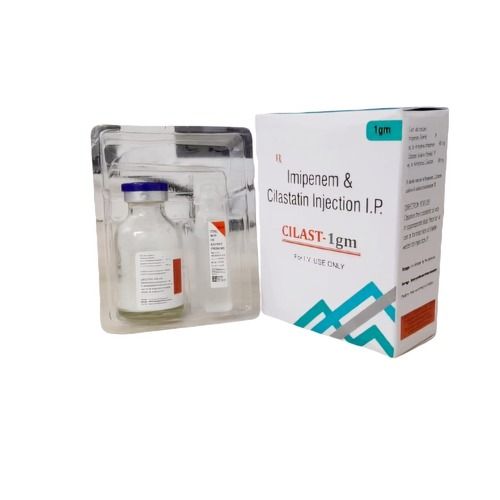
Imipenem Cilastatin Injection
Price 1059.0 INR / Unit
Minimum Order Quantity : 100 Units
Physical Form : Powder
Ingredients : imipenem cilastatin injection
Dosage Guidelines : as prescribed by physician
Dosage : as prescribed by the physician
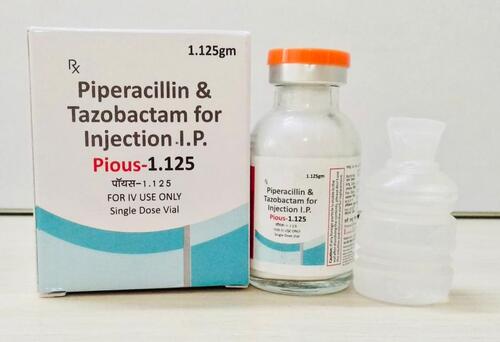
piperacillin tazobactam injection
Price 111.0 INR / Unit
Minimum Order Quantity : 100 Units
Physical Form : Powder
Ingredients : Piperacillin 1gm+ Tazobactam .025gm
Dosage Guidelines : as prescribed by physician
Dosage : as prescribed by physician
Cefoperazone Sulbactam Antibiotic Injection
Price 295.0 INR / Unit
Minimum Order Quantity : 100 Units
Physical Form : Other
Ingredients : CEFOPERAZONE SULBACTAM INJECTION
Dosage Guidelines : As directed by physician
Dosage : As rescripted physician


 Send Inquiry
Send Inquiry